I am at a stage in my illness where I am scanning the horizon and asking the following question: how can I mitigate the risks involved with raging inflammation, poor sleep, and no exercise – three hallmarks of ME/CFS and huge risk factors for Alzheimer’s disease, diabetes, cardiovascular disease, and cancer? The prospect of growing older with this disease is daunting, to say the least.
The standard advice given by doctors to maintain a higher quality of life and increase longevity is to eat a good diet, exercise, and get adequate sleep. With ME/CFS, exercise and decent sleep are mostly off the table. Diet is at least one aspect of our health we can control somewhat.
In January 2019, I started the ketogenic diet to see if pulling on different dietary levers could help mitigate some of the risks that come with this disease. In this three-part series, I explore a few road bumps I have hit along the way and discuss my approach to resolving them. Part I explores different hypotheses explaining my rise in heart rate while on the diet. Part II covers the large increase in LDL cholesterol I experienced in response to the diet and part III advocates for a new way of thinking about diet beyond the usual labels.
Why keto is good for M.E. (in theory)
I do not go much into the science of the ketogenic diet, which is covered well here and here, but the main idea is that carb restriction can mimic a fasted state in which ketones are burned as fuel rather than glucose. Ketones are fatty acids made by the liver from dietary and/or body fat. Ketosis refers to the metabolic state in which ketones are burned for energy.
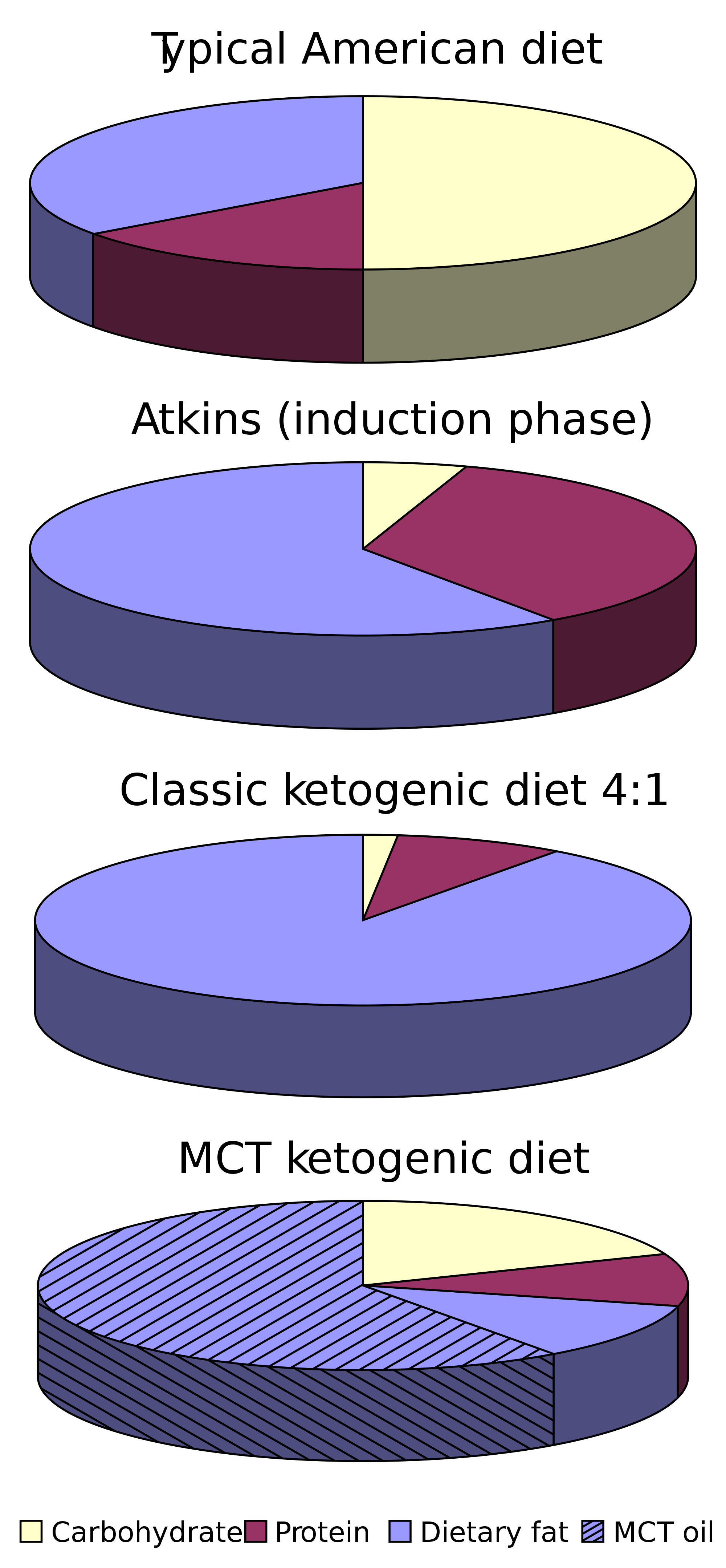
Briefly, for those not steeped in keto culture, the diet consists of consuming 70% or more of daily calories as fat, with higher caloric needs requiring more fat. Carbs are typically limited to 25 g/day (regardless of caloric needs), which is a really tiny amount, about as much as one banana. Protein consumption is sufficient but is not eaten in the quantities seen in some versions of paleo, carnivore, Atkin’s, and Wahl’s diets due to the possibility of gluconeogenesis, or the conversion of certain amino acids to glucose, which can make ketosis harder to achieve for some.
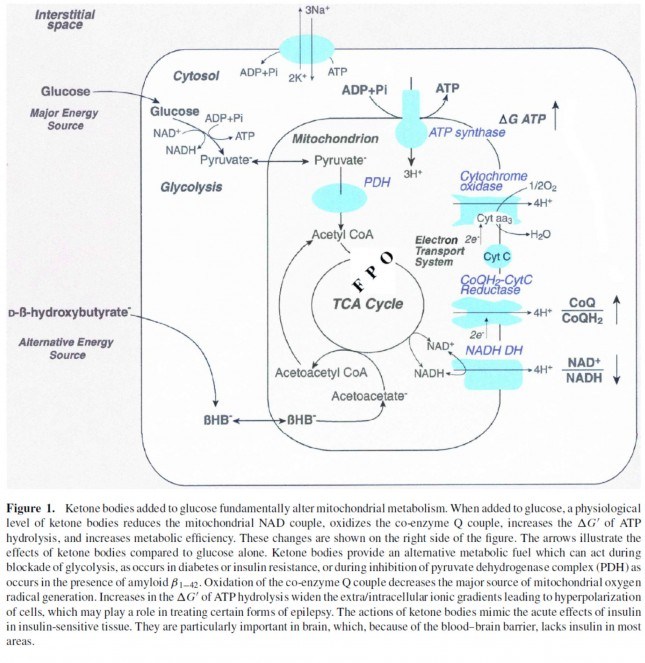
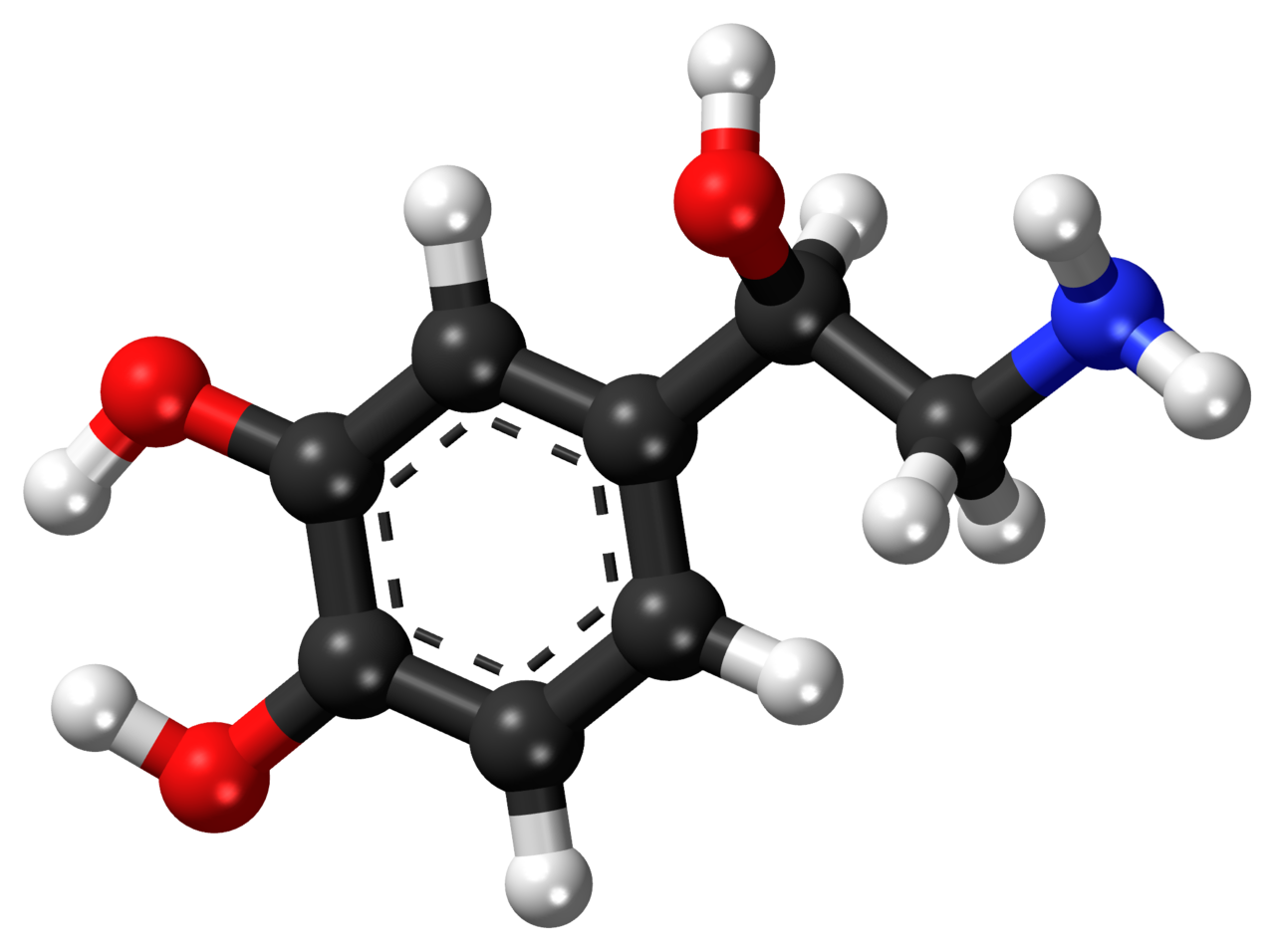
Ketones offer a brilliant solution to the energy impairments found in ME/CFS. Metabolic studies point to pyruvate dehydrogenase (PDH) as one of the culprits behind the energy production problems found in this disease. PDH is an enzyme complex that plays a key role in energy metabolism and lactate production and is the gate-keeper to the citric acid cycle (also called the tricarboxylic acid cycle, TCA or Kreb’s cycle). The TCA, combined with the electron transport chain, is a series of electrochemical reactions that convert acetyl-CoA into adenosine triphosphate (ATP) – the molecule that fuels cellular processes. Glycolysis is the metabolic pathway that converts glucose to pyruvate, which is the primary substrate for the TCA when glucose is the main fuel source. Under normal aerobic conditions, pyruvate is transported into mitochondria, where it is converted to acetyl-CoA by PDH. Because pyruvate is largely obstructed (in theory) in ME/CFS due to a faulty PDH complex, we need different ways to fuel our bodies – ketones offer one such approach.
Ketones, specifically beta-hydroxybutyrate, enter the TCA directly and are oxidized in the mitochondria for energy (ATP), bypassing PDH entirely (see below). For this reason, the ketogenic diet makes a lot of sense for people with ME/CFS by offering an alternative pathway into the TCA that does not involve the PDH enzyme complex.
I chose a ketogenic diet with six goals in mind:
- Provide a more metabolically-efficient fuel source than glucose;
- Give my pancreas a break from a burgeoning honey habit;
- Decrease inflammation;
- Improve cognition;
- Stabilize my weight; and
- Improve my cholesterol numbers.
Act I: Early days of keto
I went into the ketogenic diet pretty much blind. I started it more out of desperation than anything due to pain from inflammation and worsening brain fog. Normally, I obsess over the research before starting something new. I figured how hard could it be after following other big dietary interventions, such as the Gerson Therapy and Wahl’s Protocol? I quickly found myself on a steep learning curve (and am still on one five months into the diet). Instead of waiting until I know how it all ends up, I am going to write about my experience as it unfolds.
I could share many little tips along the way I would have known had I done a little more research before starting! For example, my advice (to myself) would be to not get caught up on the macronutrients (protein, carb, fat) at the expense of micronutrients; start low and slow if new to MCT oil (it gave me migraines and horrible brain fog); have a plan to replace fiber (psyllium and chia seeds are great); don’t over-eat protein (which can be converted to glucose); don’t eat sugar alcohols that spike insulin (even if no carbs); and consume vast quantities of electrolytes and water (more than usual for ME/CFS).

As I worked my way through these and other kinks, within days the fog started to lift. I started writing again after a long hiatus. I plowed through three novels in the first month. I woke up feeling like the Tin Man from the Wizard of Oz after Dorothy oiled his joints, not quite bouncing out of bed, but a vast improvement in inflammation. After four years of struggling with appetite and keeping weight on food started to interest me again. As I saw it, I could place check marks next to five of my goals within the first month. Hooray for keto! Goal number six is still unfolding and I will address that in its own blog post.
(This is that part of the story where the heroine is seen skipping merrily down the lane as dark clouds gather behind her…)
Act II: Hitting my first road bump on keto
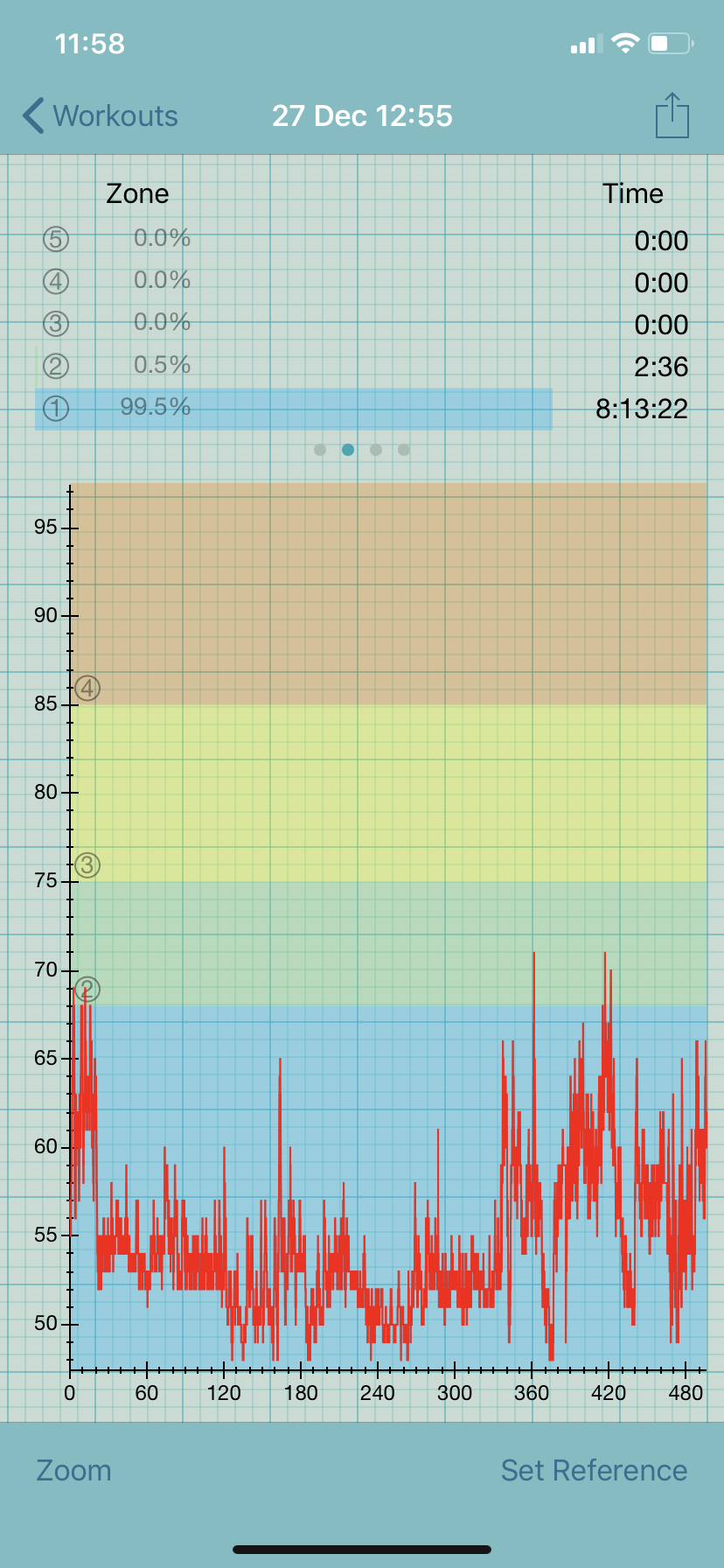
A few weeks prior to keto, I started Mestinon (pyridostigmine), a myasthenia gravis drug that is used off-label to treat dysautonomia. Within a week, my resting heart rate (RHR) dropped to the high 40s, which is my sweet spot. My postural tachycardia syndrome (POTS) virtually disappeared. I recall standing in the kitchen on New Year’s eve without exceeding my anaerobic threshold (AT), which at 69 bpm is exceedingly low. The AT places a ceiling on the amount of activity that is safe for people with ME/CFS because exceeding it causes post-exertional malaise (PEM), which is a worsening of symptoms after exertion.
Within a few days of starting keto, I saw my RHR rise 10-15 beats to 60-62 bpm. This may seem great to people with a higher resting rate but is not due to my low AT. With a RHR of 60 bpm, I spend more than 50% of my day above this threshold, even while in bed.
Adding insult to injury, my POTS has worsened since starting keto, meaning that my heart rate is in the 70-80s while sitting and my standing heart rate can exceed 100 bpm.
Given the many gains I have seen since starting keto, I figure it is worth trying to get to the bottom of this problem.
Some hypotheses to explain my elevated heart rate:
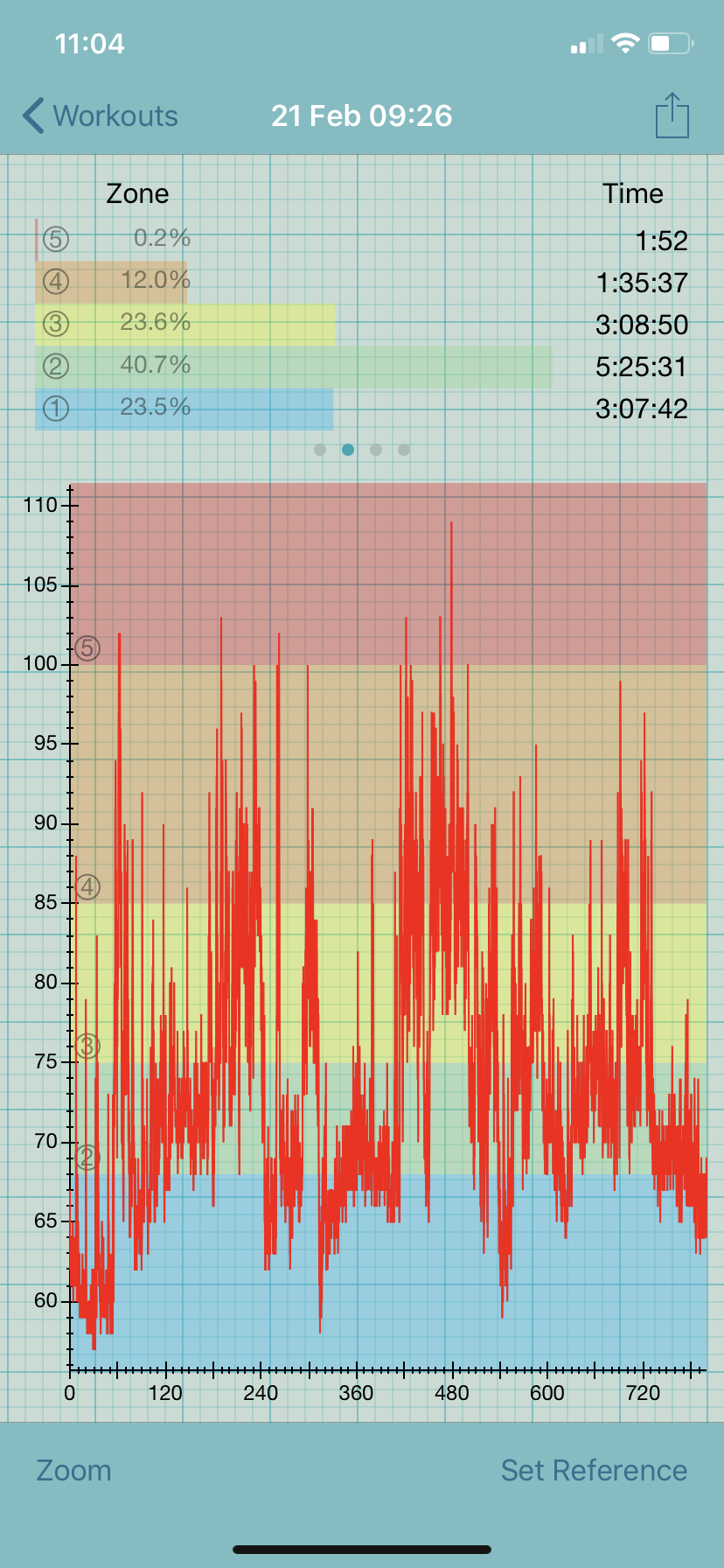
1 | This is natural variation in my disease, having nothing to do with diet.
2 | My electrolytes and hydration are off.
3 | I am eating different foods and more of the foods I usually eat only sparingly. This might be triggering a mast cell response that is causing a rise in heart rate.
4 | The change in diet altered my gut flora, which has influenced neurotransmitters that affect heart rate (a suggestion from one of my ME/CFS doctors).
5 | Since a ketogenic diet mimics a fasting state, I may be experiencing “too much autophagy” (another suggestion from my doctor).
Testing the hypotheses (N=1)
I am working through these hypotheses in hopes of finding a way forward with the ketogenic diet.
1 | Natural variation hypothesis
Having a low AT makes it very easy to get into a positive feedback loop with PEM. When I am in PEM my heart rate is elevated, which decreases the wiggle room I have before exceeding my AT, causing yet more PEM. Essentially, PEM begets PEM. Over time PEM can become chronic, also referred to as rolling PEM. I have had an elevated heart rate like this for months in the past, having nothing to do with keto.
In addition to having an elevated heart rate, my autonomic nervous system has been more unbalanced than usual since starting keto. More time spent above AT can translate to increased activity in the sympathetic nervous system. This leads to an elevated heart rate, which further exacerbates PEM. After I started keto I saw a shift toward lower heart rate variability, indicating sympathetic overdrive.
To test this hypothesis, I would need to hold all variables constant (including staying on keto) and head back to my electric wheelchair for some serious pacing. I did this in 2017 and saw my numbers, which were not too dissimilar to what they are now, stabilize. After four months of aggressive pacing (sounds like an oxymoron but trust me it is not!), my heart rate dipped back into the 40s. For a whole bunch of reasons, I cannot do that intervention just yet but am mindful that my elevated heart rate could all be background noise from being in perpetual PEM.
2 | Electrolyte hypothesis

The ketogenic diet is notoriously hard on electrolytes, which is not good for people with dysautonomia and low blood volume. Keto can cause electrolyte loss due to decreased insulin production (not as much needed when not consuming vast amounts of glucose). A decrease in insulin leads to hormonal changes in the renin-angiotensin-aldosterone system, which signals the kidneys to excrete more water and electrolytes. This needs to be managed carefully in someone with ME/CFS.
I was aware of the electrolyte problems on keto and have been eating Vitassium Salt Stick capsules like candy, but I don’t think it has been enough. The obvious first thing to do is to increase electrolyte and water consumption. I will know if I have it right when my muscle cramps disappear. I still am getting them in my diaphragm and feet on occasion, indicating there is some room for improvement. Hopefully, my resting heart rate will decrease when I have the electrolytes under better control.
3 | Mast cell hypothesis
Mast cell activation can cause havoc in the heart. In the extreme, a mast cell reaction can cause Kounis syndrome, an acute coronary syndrome which causes angina, tachycardia, and even death. I do not have Kounis syndrome and, in general, am not a patient who struggles mightily with mast cell and histamine issues as do many with ME/CFS. Still, I cannot discount this idea.
I am at a bit of a loss on how to test this hypothesis as I have not seen any clear patterns with food items and heart rate, apart from citrus and kombucha causing a transient rise of 15-30 bpm. The other thing I have noticed since starting keto is that my heart rate goes up 10-20 beats about an hour after eating and stays elevated for an hour. I have tried omitting various foods that are known either to contain histamine or aggravate mast cells, such as fermented items. I saw no difference in my heart rate in response to these measures.
I am limited in how much I can restrict as the very foods that are linked to mast cell activation are some of the same ones that help lower my LDL cholesterol. Instead of further restricting my diet, I have decided to try diamine oxidase (DAO), a supplement which helps to break down histamine (Cromolyn did nothing or me). I realize this is hardly an exhaustive approach, but if I see some encouraging signs I will continue exploring the mast cell connection.
4 | Altered gut flora hypothesis
The ketogenic diet can remodel the gut microbiome and this, in turn, might influence neurotransmitters that affect heart rate. Given that about 90% of the body’s serotonin is made in the gut by enterochromaffin cells, this seems like a possibility.
There is a growing literature on the effects of the ketogenic diet on the gut microbiome and certain neurotransmitters such as serotonin, but this is focused mostly on epilepsy. Altered levels of serotonin have been associated with a variety of diseases, including cardiovascular disease.
Testing this hypothesis would require me to revert back to a higher carb diet to see if I could nudge my microbiome back toward a pre-ketogenic state. I suppose I could run another Ubiome panel while on the diet, then another after stopping it (I have pre-keto data). That sounds pretty expensive to me! Even if I had these data it would be hard to know the possible contribution of an altered gut microbiome to my heart rate woes (a fundamental limitation of N=1 “experiments”). Moreover, if I were to introduce more carbs in hopes reverting back to a hypothetical pre-keto gut microbiome I would likely impact many processes unrelated to the alterations to the microbiome, making it really hard to assign causality.
5 | Too much autophagy hypothesis
Autophagy – which translates to “self-eating” – occurs in a fasted state, during sustained aerobic exercise, and under carbohydrate restriction (i.e., ketogenic diet). Autophagy is the ultimate form of cellular cleansing and occurs when lysosomes – little sacs (organelles) filled with enzymes that kill cells – help to rid the body of cells that are suboptimal, such as ones with DNA damage, and make way for newer, healthier cells. Intermittent fasting (and the ketogenic diet) have been touted as being beneficial for many chronic diseases, including ME, because of its ability to activate cellular mechanisms that help with immune function and reduce inflammation.
There clearly needs to be a balance with autophagy; too much or too little autophagy is not good. But how much is too much?
Although intermittent fasting has shown many benefits, fasting, or fasting done unsafely, can have some downsides, including a few that can affect the heart. Fasting has been shown to influence catecholamines, especially norepinephrine. Norepinephrine is part of the fight or flight response by the sympathetic nervous system and increases heart rate and blood pressure. Sound familiar?
Although I do not know how my pre-keto and keto plasma norepinephrine levels, my heart rate certainly has been elevated since starting the diet. As mentioned, my sympathetic drive has also increased on the ketogenic diet, as seen in lower RMSSD readings. Perhaps keto is increasing sympathetic activity through increased norepinephrine, but who knows. Apart from measuring norepinephrine levels before, during, and after keto, it would be hard to know what might be causing the increase in sympathetic drive that I am experiencing.
Act 3: Stay tuned!
My next steps are to work my way through these hypotheses, starting with electrolytes. I am limited in testing the mast cell hypotheses due to a cholesterol experiment currently underway (next blog!). I may need to up my carbs by bringing in some more fruit and sweet potato to see if that does the trick.
In the end, I may conclude the ketogenic diet is not for me. However, I am loath to stop doing something that has helped in so many ways. I have seen many gains in cognition, appetite, weight stabilization, and energy. However, one of the most fundamental aspects of managing ME/CFS is pacing – keeping heart rate below our anaerobic threshold. Since starting the ketogenic diet this has been nearly impossible for me.
I can’t say where this will all end. I’ll report back when I have my answer!
UPDATE: Not so long after I wrote this piece my heart rate normalized, or at least went back to it’s usual wonky patterns. I added 200 mg of magnesium and that resolved all of my cramping and may have contributed to the resolution of my chronically-elevated heart rate.
I also think that it took several months for me to become adjusted to being a fat burner, despite the fact that I had excellent ketone levels early on. I am happy to report that my keto nausea has also resolved. I can’t rule out the natural variation hypothesis either as my body does experience prolonged crashes – on the order months – in which my sympathetic drive is stuck in the on position.
I may never know what was driving my elevated heart rate, but I can rule some of the hypotheses out, namely that it was due to changes in my gut flora that were influencing neurotransmitters. I also am skeptical about mast cell reactions in response to dietary changes. I haven’t changed much in my diet, apart from relaxing carb intake as long as my beta hydroxybutyrate (the ketone I monitor in my blood) levels are at or above 1 mmol/L.
Putting all of this together I think I just needed time and magnesium. So glad I stuck it out and am not left to wonder why I had this reaction.

Caroline do you use breathing techniques to influence your HRV ? I’m a scientist too and investigate voluntary augmentation of the parasympathetic nervous system in states off illness.
I also know a bit about keto amd NE but not more than you ☺
I have reduced orthostatic symptoms and improved function through various methods. Contact me I’d love to chat. “Claire hrvinptsvshcs Winzar” on fb.
LikeLiked by 1 person
Caroline- you are awesome. Thanks so much for sharing your journey, and doing so in such a helpful way for others to learn from your experieces. You are a great teacher. I appreciate (and like) the goals you have for this experiment, the hyposthesis you have, and very much like your reporting out along the way. I send cyber hugs of support. Wishing you the very best.
LikeLiked by 1 person
Caroline, thank you for your story. I have had similar frustrations with an elevated heart rate on keto, especially after a meal. At night, my resting heart rate is between 45 and 50bpm, but after a meal, it will hover around 100bpm for an hour or 2. If i eat more sodium, my blood pressure will increase to 140/90, so I now eat a low sodium diet, which some youtube keto experts will blame for my tachycardia. I drink Dr.Berg’s electrolyte drink 3 times a day to get enough potassium it seems, but it sometimes causes heartburn or nausea. I workout often, lifting weights daily and running more than 40 miles per week. Don’t know if that has made my keto adaptation more challenging, since I’ve been keto for more than a year now, and just turned age 60. Any advice is welcome. Thank you
LikeLike
Thank you for reading my blog! Sorry to hear you have an elevated HR in response to keto – and that your HR goes up so much after a meal! Does it do that regardless of what you eat? I know that certain foods will raise my heart rate over others. I also struggle to get enough potassium due to nausea/GI upset. that is great you can work out! Oh, how I miss that!
LikeLike
One other little tidbit to pass along…I was listening to an amazing podcast on blood pressure and salt and how it isn’t salt per se that causes hypertension but rather serum osmolality. Turns out that drinking a glass of water before or with a salty meal helps! And, apparently, sugar can cause a rise in BP! But being keto thankfully we don’t need to worry so much about that. https://peterattiamd.com/rickjohnson/
LikeLike
I believe there is a small subset of people who have seen an increase in rhr with low carbohydrate eating. I am one. Six years of lchf has seen my rhr permanently increased from 68 to 78. Everything else is fantastic. I’m a lmhr too.
LikeLike
Wow! So your RHR never dropped? Mine FINALLY did after about 5-6 months and I am back to my usual pattern of having a really low RHR when in a parasympathetic crash and higher RHR when sympathetic dominant. I recently tested the waters with some sugar and paid the price with massive inflammation. If ever there is a potent motivator to avoid sugar, inflammation is it for me. Ahh…a fellow LMHR…I haven’t tested in a while, but last time I did my numbers were looking a bit more respectable – with LDL-c in the 200 mg/dL range (down from 330 mg/dL). A combination of diet and 10 mg of Zetia seemed to really help. I know some don’t worry about the sky-high LDL-c but I figure I still have plenty to spare 😉 Thanks for reading!
LikeLike
Thank you SO MUCH, Caroline for all the info you have shared in this article and others. I look forward to read more of your writing. it is very informative and helpful.
LikeLike
So pleased you found it helpful! Did you also have an elevated heart rate when you started keto? Mine eventually settled…
LikeLike
This is an interesting topic. I am on a LCHF diet for 4 years…with ketone-levels between 0.1 und 2.0 …10 days ago, for various reasons, I went 100% keto with less than 20g CHO a day. Now my heart rate is +8 during the night, die HRV dropped by nearly 50% and doring excercise my heartrate is +20.
I feel grate, but the oura ring does not like this development at all 😉
I think this is normal for the first weeks of keto…but it is fascinating that this happens after 4 years of LCHF!
Kind regards from Berlin,
Robert
LikeLike
Wow! How fascinating – after four years of LCHF, your body still responds to a drop in carbs. That pesky Oura Ring, what does it know anyway, lol? It does seem to be transient. So interesting how many things improve while LCHF but other things, such as sleep and HRV, take a hit. Thanks for reaching out!
LikeLike
Just want to thank you for posting this, as I’m having the exact issues and needed some reassurance from someone experiencing the same
LikeLike
Hi Ryan – Do you have an elevated heart rate in response to keto? If so, I will be so curious to hear when it comes back down. This is a tough one for people with ME/CFS because it leaves us with less heart rate reserve before hitting our anaerobic threshold. I seem to be back down in the 40s again – giving me so much more wiggle room. Best to you!
LikeLike