Many people who go on the ketogenic diet see incredible health gains only to discover their LDL cholesterol (LDL-C) has skyrocketed. Rightly so, many people resist giving up their low-carb lifestyle due to its many benefits. A deep distrust of doctors and pharmaceutical companies, not to mention decades of farcical advice on nutrition, leads people to wonder if keto and their high LDL-C is truly the problem it is made out to be. In this post-truth world, it is not hard to find sources that will confirm any viewpoint.
Where do you turn if you want to continue keto but do not want to risk cardiovascular disease down the road? Who should you trust? There is so much conflicting information and confirmation bias surrounding cholesterol. Doctors do themselves no favors when they refuse to be in partnership with their patients, making it hard for people to forge an individualized treatment plan. Cardiologists are best positioned to help patients assess their risk but the usual 10-minute appointment slot does not allow for more nuanced discussions.
Treatment guidelines from the American College of Cardiology (ACC) and the American Heart Association (AHA) also put up roadblocks for patients trying to understand what is driving their high LDL-C. The 2019 guidelines stipulate that patients with LDL-C ≥190 mg/dl should “use a high-intensity statin without risk assessment“. This means that patients with high LDL-C are simply sent off with a prescription for a statin without further analysis. Insurance companies know this and can limit more advanced lipid testing, claiming it as “experimental”.
Assessing risk is key when you are a hyper-responder and have a complicated health picture, as I do with myalgic encephalomyelitis (M.E.), or if you do not want to take statins. In this piece, I present two lipid models – the lipid model of atherogenesis and the lipid energy model – as a foundation for assessing risk. The lipid model is widely accepted by researchers and physicians and has been refined over several decades. The lipid energy model is largely untested and was developed only recently in response to the increasing popularity of low-carb, high-fat diets (LCHF), such as keto and carnivore.
The lipid model of atherogenesis
Atherosclerosis is the leading cause of mortality in the United States and much of the developed world and is a complex product of genes, environment, and lifestyle choices. That much most everyone seems to agree on.
Cardiology is an ever-changing field with plenty of its own internal controversies. Increasingly, the predictive value of LDL-C in heart disease is called into question by both lipidologists and keto enthusiasts, but for different reasons. It is important to understand where there is widespread agreement and which aspects of the lipid model are up for debate.
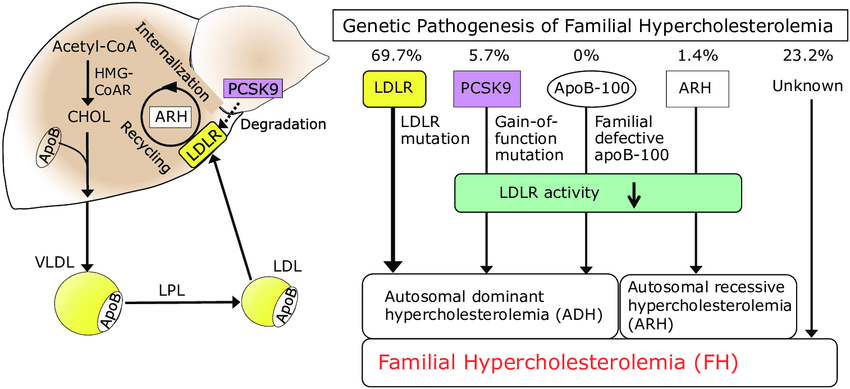
According to the lipid model, atherogenesis occurs when lipoproteins make their way through gaps between the endothelial cells lining vessels and arteries and end up in the sub-endothelial space. It is a numbers game: the higher the LDL particle burden in the bloodstream, the greater the chance lipoproteins will squeeze through the endothelium.
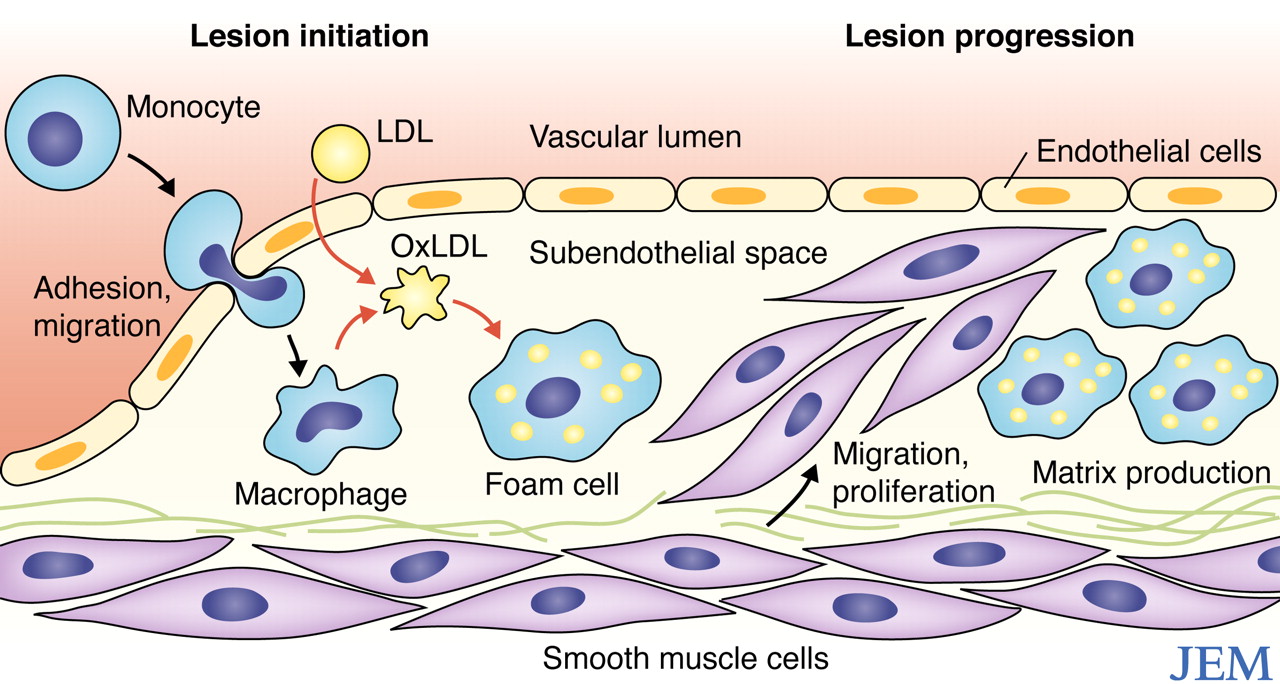
When an LDL particle ends up in the sub-endothelial space it can become oxidized. This triggers an inflammatory response in which monocytes – a type of white cell – migrate through the endothelium and proliferate as macrophages. Damaged endothelial cells release cytokines which signal the migration of smooth muscle cells that form a fibrous cap called an atheroma. Over time, calcium can build up causing the “hardening” of the arteries. Thus, atherogenesis is a complex response to injury.
With time (can take decades), new plaques form and the inside of the artery may begin to narrow. When plaques rupture they cause a heart attack or stroke.
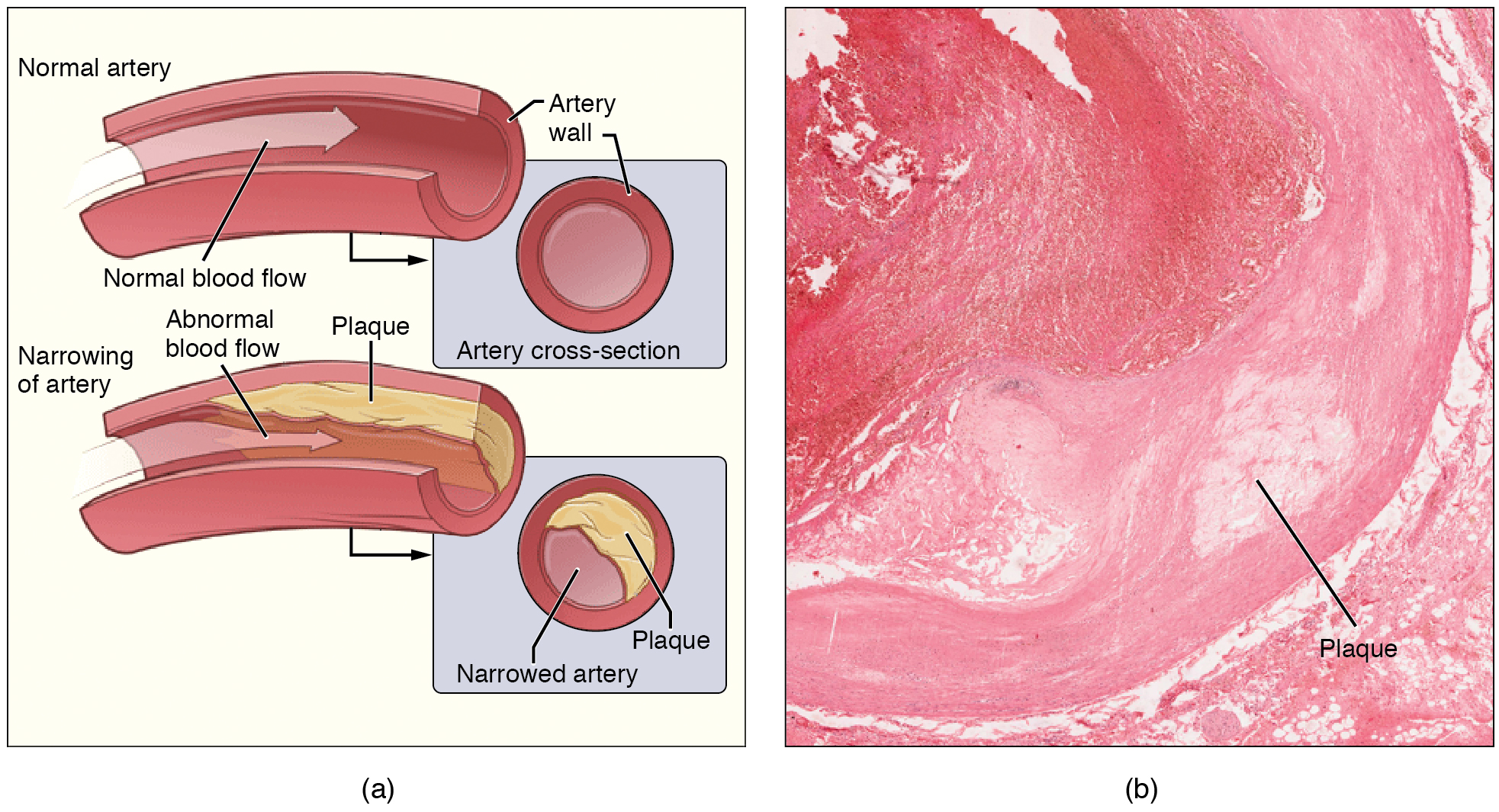
Despite overwhelming evidence in support of the lipid hypothesis, the association between LDL-C and heart disease is not as clear as doctors make it out to be, at least for a subset of people. A 2009 study showed that 50% of heart attacks were not associated with elevated lipids, prompting calls to lower the threshold for statin treatment. At the same time, some people with dangerously-high LDL-C are perfectly healthy.
Metabolic health is the X-factor that seems to determine how problematic LDL-C is, an idea that many keto enthusiasts hope to be true. Lipidologists agree that poor metabolic health can accelerate atherogenesis, but do not necessarily agree that metabolically-healthy people are immune to heart disease.
In my next post, I drill into better predictors of heart disease related to LDL-C, including LDL particle count (LDL-P), LDL size distribution, and ApoB.
The lean mass hyper-responders (LMHR)
When I received my test results this past spring I did what anyone would do: I consulted the internet. Fortunately, I got my first break when someone on Facebook mentioned that I might be a lean mass hyper-responder (LMHR) and suggested that I head over to Cholesterolcode.com. This thrust me into a bizarre and fanatical world of others just like me who went on the ketogenic diet and saw a sharp rise in LDL-C.
What on Earth is an LMHR, you ask? LMHRs are people who are very lean, typically athletic, with fairly normal lipid numbers before starting keto. In response to a high-fat diet, they see their LDL-cholesterol skyrocket. However, their other markers are excellent, with HDL-C >80 mg/dL, triglycerides <70 mg/dL, and high insulin sensitivity. Other than the high LDL-C, they are otherwise healthy people.
Despite being lean and a hyper-responder, I don’t quite fit the LMHR profile because I had dyslipidemia before keto and my trigs are a little too high. While I have excellent insulin sensitivity, as a person with M.E. I have a number of metabolic impairments (topic for a future blog) that could be influencing my dyslipidemia. Still, I have many LMHR elements.
Open up it’s me, Dave, I got the stuff… (I couldn’t resist a Cheech and Chong reference here)
At the center of the LMHR world is Dave Feldman, a software engineer cum amateur lipidologist. He is a fearless truth-seeker who regularly takes on prominent cardiologists and lipidologists on Twitter. Not too dissimilar to the anti-vaccination debate, he has uncovered questions for which scientists have yet to provide compelling answers, leaving room for the idea that high LDL-C is not a problem while on keto as long as other biomarkers are good. I have a lot of respect for Feldman, but his hypotheses are untested at this point.
When I joined Feldman’s LMHR group on Facebook I found a community of people following ketogenic and carnivore diets. The members tend to be highly suspect of card-carrying scientists and any research involving cholesterol-lowering drugs such as statins, claiming they do more harm than good. Still, the unease you feel among many in the group is palpable and some have LDL-C much higher than mine!
Feldman’s lipid energy model
Feldman’s lipid energy model goes something like this. When following a low carb/high fat diet (LCHF), such as keto or carnivore, in which fat is the primary fuel source, there is a greater need to transport triglycerides to muscle cells for energy, especially in people with a low BMI (the lean mass part) and depleted glycogen stores (which happens on keto).
He uses the following analogy to explain his model. You might notice there are a lot of pizza delivery trucks (aka lipoproteins) out on the road, presumably because there was a massive increase in both the supply (fat) and demand (energy as triglycerides) for pizzas. If you were to pull over the pizza trucks many of them would likely be empty because they dropped off their pizzas already. Feldman talks about VLDLs (the pizza trucks with pizzas onboard) in much the same way, claiming that the reason why we see so many more LDLs (empty pizza trucks) on an LCHF diet is that there are more triglycerides to be delivered via VLDLs, which then go on to remodel into LDLs. According to Feldman, the increase in LDLs is merely a response to the increased need for shuttle fatty acids to tissues.
Unlike the lipid model, the energy model does not rest on the causality of LDL-C in atherogenesis, bucking decades of research and advice from cardiologists. If you are lean, have high HDL-C, low trigs, low inflammation and are insulin sensitive – which are essentially markers of metabolic health – high LDL-C is not a problem, according to the model. In fact, LMHRs view high LDL-C as protective and a sign of a healthy lipid trafficking system.
Say what? But isn’t having too much LDL-C bad?
I get it. I would love nothing more than to believe my high LDL-C is not a problem and may even be beneficial. When I started keto I saw many gains and am resistant to giving it up. Many LMHRs have seen even greater benefits, walking back type II diabetes, losing weight, and feeling more energetic than when they were eating the Standard American Diet. Many proudly tout their LDL-C numbers, often shaming others who question whether their LDL-C is harmful.
Personally, I have no interest in cherry-picking the data to support a view that may, in the end, kill me. As a Ph.D. scientist, I am trained to be skeptical until convinced otherwise. I mean no disrespect to Feldman and his followers but remain unconvinced for now merely because the data to evaluate his hypothesis adequately do not exist currently. For that same reason, I also remain open-minded. Even Feldman himself is careful to say that he may be wrong in the end, but he is cautiously optimistic.
Some outstanding issues (in my mind) with the energy model
Feldman does not deny that LDLs are involved with heart disease, but he thinks that high LDL-C (and LDL-P) is not the problem. According to him and other LCHF proponents, the epidemiological data (which is observational) do not show increased mortality for those with high LDL-C, trigs <70, and HDL-C >80, i.e., the triad. Most lipidologists agree that atherosclerosis is impacted by many factors beyond lipoproteins, but that doesn’t diminish their role in cardiovascular disease. As Peter Attia puts it, lipoproteins are necessary, but not sufficient, for atherosclerosis.
High HDL may not protect you. Many LMHRs believe that high (but not too high) HDL-C protects against heart disease, presumably because of the role HDLs play in clearing excess cholesterol from the body. In the last decade or so, HDL-raising drug trials and genome-wide association studies (GWAS) have debunked the idea that HDL is as protective as once believed and most cardiologists now will rebuke the idea that high HDL-C makes high LDL-C alright to have. Instead, HDL, along with triglycerides, is likely an indirect measure of metabolic health. High HDL-C might not protect against heart disease, but low HDL-C and high triglycerides could certainly contribute to it. Recent research focuses on triglycerides as a causal factor.
For a deeper dive on HDL check out this podcast with Dr. Ethan Weiss (a UCSF preventive cardiologist) interviewing Dr. Sek Kathiresin (cardiologist and CEO of Verve Therapeutics). Also, Attia’s 3rd interview with Dr. Tom Dayspring does a deep dive on HDL.
Cardiac events are not soft endpoints. LMHRs like to point out that many trials involving lipid-lowering drugs do not show an appreciable reduction in cardiac deaths and all-cause mortality. There are a few issues with this reasoning. First, short-term drug trials are not designed to use cardiac and all-cause mortality as the outcome – this would require at least a decade or more because of the longer timescale for atherosclerosis. Despite this limitation, short-term trials for a newer class of drugs called PCSK9 inhibitors showed dramatic reductions in revascularization events (i.e., angioplasty and stent) in a three year period. Feldman would call this a soft outcome.
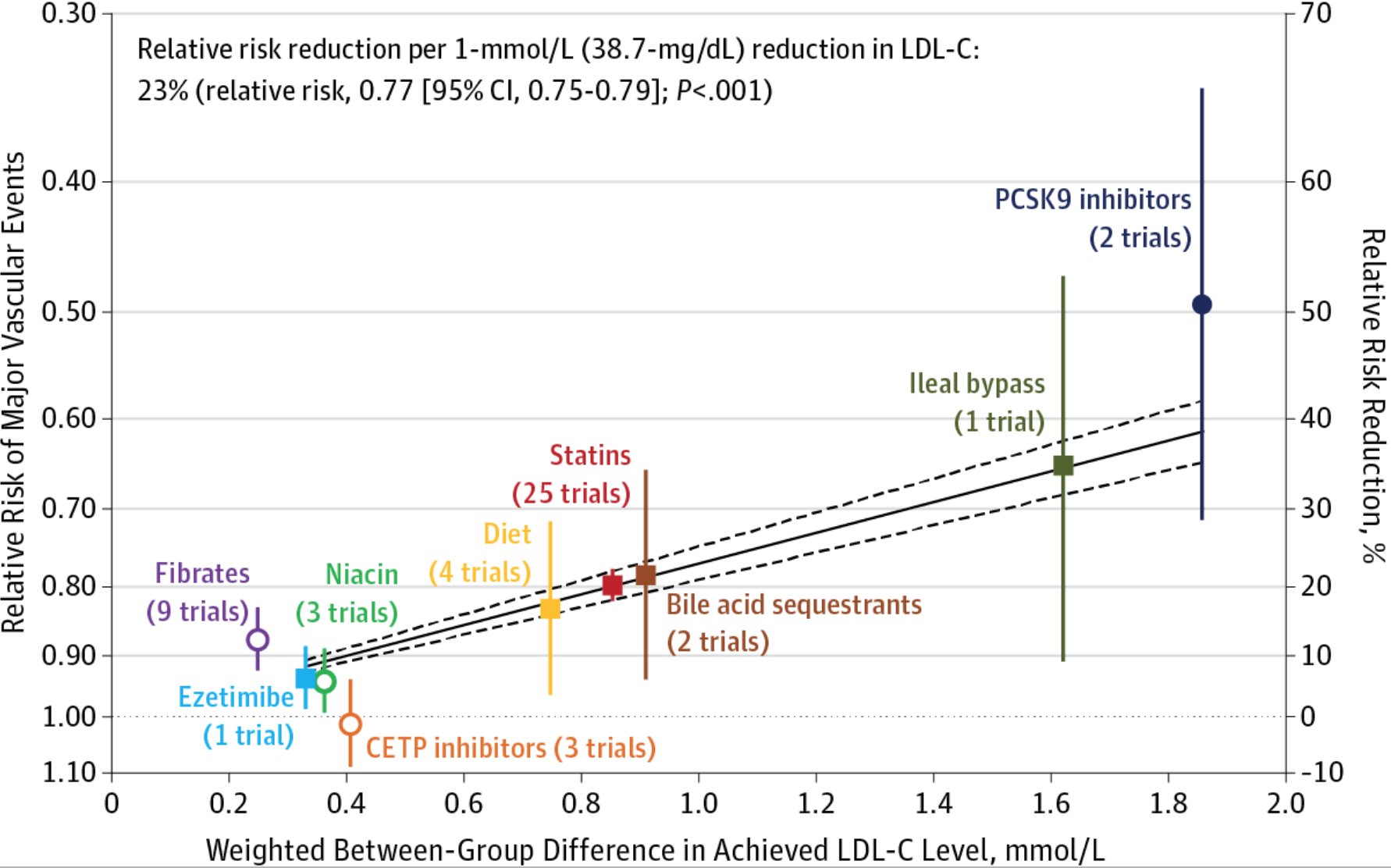
Cardiac events that don’t result in death can greatly impair the quality of life. Older physicians tell stories about how, before statins, ERs would see multiple heart attacks each night, many with poor outcomes, including death. We have come a long way in preventing and treating heart attacks since those days, thankfully, but these events still carry consequences.
LMHRs argue that life expectancy with familial hypercholesterolemia (FH) – defined as LDL-C >190 mg/dL – is around the same as it is in the general population, so LDL-C can’t be all that bad. Well, that isn’t exactly true. Mortality from cardiac events is much higher in FH, but fewer die from cancer and other diseases common later in life, assuming people make it that far. It is like saying do not worry about the heart attack or stroke that could kill you today because you will not get cancer when you are older! Many assume that it is the high LDL-C conferring the benefit later in life, but it could be due to other (beneficial) genetic variants linked to FH having nothing to do with cholesterol. Likewise, correlated factors other than high LDL-C could contribute to elevated cardiovascular risk in FH.
Why don’t all lean, metabolically healthy people see a rise in LDL-C on keto? About 20-30% of people on keto see a massive spike in cholesterol. To deny that such a hyper-response has no genetic basis seems pretty absurd (but I get that LMHR might not have FH). If this were true, why don’t all lean people see their LDL-C go through the roof when they start keto, as implicated in the energy model? With over 2000 known genes associated with cholesterol metabolism and likely many more, we cannot categorically state that the hyper-response to keto/carnivore has nothing to do with genetics. There are likely several common genetic variants at play.
VLDL and triglycerides would show the opposite pattern of what Feldman predicts for fat burners. At the heart of Feldman’s energy model is the idea that increased LDL-C on keto is simply a response to a greater need to transport more fats. Thus, it is an adaptive response to being a fat burner, according to the hypothesis. However, in insulin-sensitive people, fewer trigs are exported from the liver in VLDLs and their residence time is less, not more, as would be required to remodel a VLDL to an LDL. Further, about 40% of LDLs are produced de novo, meaning they do not originate from the VLDL line, though this percentage is not known for keto-adapted people. In other words, not all LDLs represent empty pizza delivery trucks.
According to lipidologist Tom Dayspring, if an LMHR were cranking out more VLDLs in response to increased fat consumption you would also see much higher serum triglyceride levels. If anything, keto normalizes triglycerides.
Even if the energy model were correct, it still does not necessarily get LDL off the hook. Just because we do not eat a lot of sugar and are metabolically healthy does not necessarily mean high LDL-C is OK. Keto only goes so far in mitigating some of the risk factors. Still, we cannot say either way if the higher LDL-C burden while on keto translates to increased risk, as central lipid dogma suggests.
Just because low LDL-C may carry health risks does not mean that sky-high LDL-C is good. Another fallacy is that if some LDL-C is good, more must be better. Feldman and others have been looking at some of the epidemiology data sets and are not finding evidence of higher mortality rates with higher LDL-C when HDL-C and triglycerides are good. However, these data sets simply do not have enough people with really high LDL-C levels. Sky-high LDL-C is fairly rare in the population and getting large enough samples for meaningful analysis is tricky. Likewise, just because low LDL-C carries risks (e.g. hemorrhagic stroke in post-menopausal women) does not mean that really high LDL-C is a good thing. There is likely a sweet spot, a u-shaped curve perhaps. Too little or too much cholesterol is not good, but intermediate levels may confer advantages.

Coronary artery scans only tell you where you’ve been, not necessarily where you are headed. LMHRs use the coronary artery calcium (CAC) scan as a primary measure of risk, especially if they have a history of eating the Standard American Diet and/or smoking. The CAC scan provides a score of how much arteries have calcified.
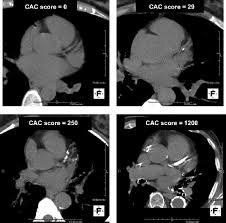
The problem with CAC scans is that it can take decades for calcified plaques to develop, long after soft plaques have appeared. Soft plaques are riskier to have but a CAC scan cannot detect these. The test can be helpful if you are young and you a high score (which might suggest intervention), but most young people will have a CAC score of zero, making it a useless test until you are in your 40s, 50s, or older. At age 51, I have a CAC score of 0, but with my LDL-C (and particle count) being so high I have no idea if I have soft plaques. The only way to know if plaques are developing is with CT angiography (CTA), but this test that is hardly ever ordered and insurance rarely pays (same with CAC test, which is very cheap – mine was $99).
Here is an informative video about how the CAC scan can be used to assess cardiovascular risk.
Finding middle ground
LDL-C is a well-established independent risk factor when looking at the general population, a finding supported by multiple lines of evidence. However, at this point, lipid researchers and LMHRs cannot say with certainty – either way – what elevated LDL-C (and LDL-P) means for those on an LCHF diet. The epidemiological studies, such as the Framingham Heart Study, are not very helpful because they do not stratify by diet and metabolic health (though HDL and trigs offer a proxy).
Instead, we need randomized controlled trials (RCTs) that evaluate what happens when people become fueled by fats. This would allow for specific hypotheses to be tested (see this excellent blog by Dr. Spencer Nadolosky on possible mechanisms). We need genome-wide association studies (GWAS) to help identify genetic polymorphisms related to the LMHR phenotype. In his Best Known Method podcast, UCSF cardiologist and researcher Dr. Ethan Weiss does an excellent interview with Dr. Ron Krauss – a legend in cardiology – in which they discussed the need for kinetic studies involving lipids and nutrition to better understand cardiovascular risk while on keto. This would be fairly straightforward to do. Such data would help us reconcile the two lipid models and determine whether some different rules might apply to LMHRs.
It may be that Feldman is right in the end, even if some of the mechanisms proposed do not pan out. He has been challenging the cardiology establishment to provide evidence that LMHRs are at greater risk of cardiovascular disease. Cardiology researchers like Weiss and Krauss are taking notice. Feldman is seeking a big tent and I hope he finds one. People following keto deserve better care and need support, especially given that many people are on the diet to improve their health.
The bottom line is that we are occupying a grey space that currently cannot be addressed due to the paucity of data. In the end, people on keto with high LDL-C have a tough choice to make. We know that cardiovascular disease occurs over decades. People who forgo interventions to reduce LDL-C will not know the consequences of that decision until it might be too late. It is still possible to be on keto and lower LDL-C through dietary changes. Some may decide to add in some more carbs, quit the diet altogether, or take a low-dose statin or other lipid-lowering therapy in conjunction with an LCHF diet.
All healthcare decisions are personal ones. I encourage readers with high LDL-C to explore some of the resources here and make up your own mind. I respect the different paths people walk and hope you will afford me the same courtesy as I work my way through this important issue.
It is important to remember that this field is rapidly changing; what you read here now could easily be outdated in the very near future. To borrow a phrase from Peter Attia, all facts have a half-life. But as Ethan Weiss says, you will be hard-pressed to find a physician or researcher who thinks the lipid hypothesis is wrong.
This post builds on two previous ones:
Keto and ME/CFS (Part I): An elevated heart rate
Keto and ME/CFS (Part II): The cholesterol hyper-responders
Like what you are reading on my blog? Please consider following me and sharing my work! Thanks so much!
Wow. Great overview and analysis. I hope this is read widely among people interested in heart health as well as our ME community. Cardiology has done wonders for so many–my father living to 91 with a quadruple bypass and a few stents a long the way, while his father died at age 69–so I am grateful for the “standard care” that was developed over those decades. But the one-size-fits-all approach with statins has definite limitations; they certainly hurt my immune system in the18 monty’s I was on them, and I’ve never recovered though I stopped taking them fourteen years ago. I’m grateful to you for embracing and elucidating emerging theories. If I had felt better during the 90 days of my keto experiment a couple of years ago, I may have stuck with it. Because of my own cholesterol profile, similar to yours in many ways, I was kind of relieved to go off of it too. I remain curious, and hopeful that this is a healthful path for you now and for a few more decades at least.
LikeLike
Hi Caroline — I’m quite sad to have only just now read your excellent article on my energy model, the LMHR profile, and your analysis. I thought it was very thoughtful and well researched. I really want to emphasize few people outside the low carb community truly take the time to understand the proposed lipid energy model (including lipidologists) and I think you come very close.
When I get a chance and a bloc of time to follow up questions, I’ll tweet this article with my regards to your craftsmanship. (I’m currently in the middle of this monstrosity — CholesterolCode.com/documentary — so I have to find odd gaps for social media right now)
Also — feel free to reach out to me via email if you have any further questions. Cheers!
LikeLike
Thank you for your incredibly kind and generous words – I am so honored you read by my blog! As you can see, your work has been invaluable to me and helped me to find a way through my own cholesterol journey. I am deeply grateful for all of your efforts to call attention to the LMHR phenotype and that CVD risk might not be what it would otherwise seem. I am so excited about your citizen science research and new documentary and hope that they open up better approaches for assessing risk. I deeply appreciate and respect how you have been able to move the needle on this conversation and can’t wait to see what comes next.
Your work, along with my own research, gave me the confidence to present a case against taking statins. I am very fortunate to have cardiologists who acknowledge that we don’t have good data on the LMHR phenotype and that high LDL-C in an otherwise metabolically-healthy person might turn out to be OK. I remain cautiously optimistic. Thank you, again!
LikeLiked by 1 person
Thank you for very kind words.
I’m actually surprised regarding your comments on confidence with the higher LDL-C. I had the impression you might prefer to scale back from the LMHR phenotype levels. And — as you know I stress regularly in the FB group — I think it’s important everyone come to their own conclusion and be respected for it (in other words, I’m not saying this in any leading way 🙂 )
Also, I’m going to tweet this article shortly, so I wanted to give you a heads up. Again, it’s probably one of the best and most thorough articles discussing both the summary of the model, arguments for the convention science, and a bit of sociology with the many respective communities involved. Truly excellent work!
LikeLiked by 1 person
My views are constantly evolving. After I wrote the article I had the opportunity to see a lipidologist at Stanford, which put to rest some of my concerns about having high LDL-C. I went in fully expecting them to suggest a PCSK9i, but they were fearful about how my immune system might react after injecting a monoclonal antibody. They said my CAC of 0 was a gamechanger and that they did not see a reason for treatment, but suggested I try a low dose of Zetia. If Stanford lipidologists are cool with using CAC scans every 5 years to guide treatment decisions I figure that is some huge progress.
I guess I would say I am in the middle of the road on all of this. I learned that I am a hyper-responder to saturated fats and was able to drop LDL-C by 100 points. I am comfortable with my LDL-c being ~200 mg/dL as long as my CAC stays low. I can easily achieve this with diet, Zetia, and berberine. I am fortunate in that I do not have doctors who are pushing drugs on me.
As an academic scientist, I guess I am prone to look at the weight of evidence and my article was mostly me thinking out loud, weighing your model and the lipid model. As you say, and the CSF will address, the evidence simply is not there to fully evaluate the question of whether high LDL-c is potentially problematic in someone who is otherwise very metabolically healthy and who does not have an underlying inflammatory condition. Your work will help address that.
Thanks for the heads up about posting my article – I am guessing that many will not appreciate my sentiments! But we are all works of progress – just as you keep evolving, so do my views. Truth is the most important thing to me – and to get to it we need to be able to refine our views over time. I see you doing that constantly and this is why I am such a big fan of your work.
LikeLiked by 1 person
Hi Caroline, good article.
I generally support the lipid energy model, because ApoB and LDL are elevated – to very high levels – in lean, healthy people when they fast. No-one thinks fasting causes atherosclerosis.
I diverge somewhat from Dave’s explanation (and am more in line with yours) when it comes to the reasons for higher LDL – we can see that LDL-raising saturated fats strip the TGs from ApoB in the liver and promote their oxidation, causing the ApoB to be released in a low-TG form.
But bear in mind that these saturated fats that raise LDL are not associated with CVD – eating more vs less of them in the normal diet does not increase risk.
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6451787/
I think this – as a modification, rather than contradiction of Dave’s explanation – will have most explanatory power, if keto and fasting replicate it in some way (by causing even more SFA to be oxidised in liver).
A few years ago I read through all the papers I could find from the Brown and Goldstein era of cardiology, when atherogenic dyslipidemias were being categorised.
I could not find a single description – not one case – of an atherogenic dyslipidemia with low TGs (defined as <100) and only one of 6 categories that could present with normal TGs as well as high.
As for genetic determinants of LMHR, for a start, if people who do not respond in this way are in the minority in the LMHR population then it is these people who we need to explain genetically, not the LMHRs, who have the normal response! After all, it is the non-LMHRs who are having the response that obese, type 2 diabetic, and MetSyn people would normally have to ketosis.
Atheroscerosis is a lipogenic pathology and requires a lipogenic mileu. Much of the cholesterol found in plaque is synthesized there, or is only retained because insulin is too high. It merely stays in the blood stream in LMHR because other cells don't want it – macrophages, hepatocytes, adipose all have reduced cholesterol requirements when we aren't growing bigger..
LikeLike
Thank you for reading my post! I agree, it is easy to chase your tail in circles with saturated fats. I know that I am a hyper-responder to saturated fats given my own N=1 experiments. But, as you point out, does that map onto CVD? In my case, not yet (and perhaps it never will). I have settled on eating a diet lower in saturated fat, which has brought benefits in other areas, mainly by making me pay more attention to nutrient density. I don’t feel comfortable completely ignoring LDL-C but also am ok with having higher cholesterol as long as I am eating a really clean low-carb diet and have a zero CAC score. So far, so good! Thanks again for dropping by! Best to you on your cholesterol journey 🙂
LikeLike
Thanks Caroline – this is a mystery, but I think one of the keys to understanding will be finding the reasons for the differential response to saturated fat, fasting, or ketogenic diets between the metabolically healthy and the rest.
Not just why is there a cholesterol rise in these situations, but why is the cholesterol rise in these situations vs none so often an indication of fitness?
I will share my experience of CFS/ME – I had this condition in the “Tapinui Flu” era (early 80’s) in New Zealand, my GP Dr Campbell Murdoch was one of the first to study the condition. I found B12 helpful but not much else.
At some stage, after many years, it went away, and I think the reason is that I became addicted to amphetamines, injecting ritalin or benzedrine. So I first didn’t eat, and then when I did would force myself to eat nutrient-dense food – usually steak, potatoes, and greens with butter. Which was different from the breaded and sweet stuff I normally chose. So I was probably in ketosis much of the time, intermittently fasting, and avoiding plant proteins and seed oils.
In any case I was very lucky that when all the drugs, and the Hep C I had picked up along the way, were finally out of my system I had reasonable use of my body’s energy systems.
Don’t try this at home kids…
LikeLike
Dear Caroline, please have a look at the latest video on youtube with doctor Vladimir Subbotin about the real anatomy of coronary vessels, they are really different from other artheries and the lipid ipothesis of atherosclerosis does not fit this anatomy. Regards.
LikeLike
It’s been on my list! Thanks for the reminder!
LikeLike
Thank you!
LikeLike